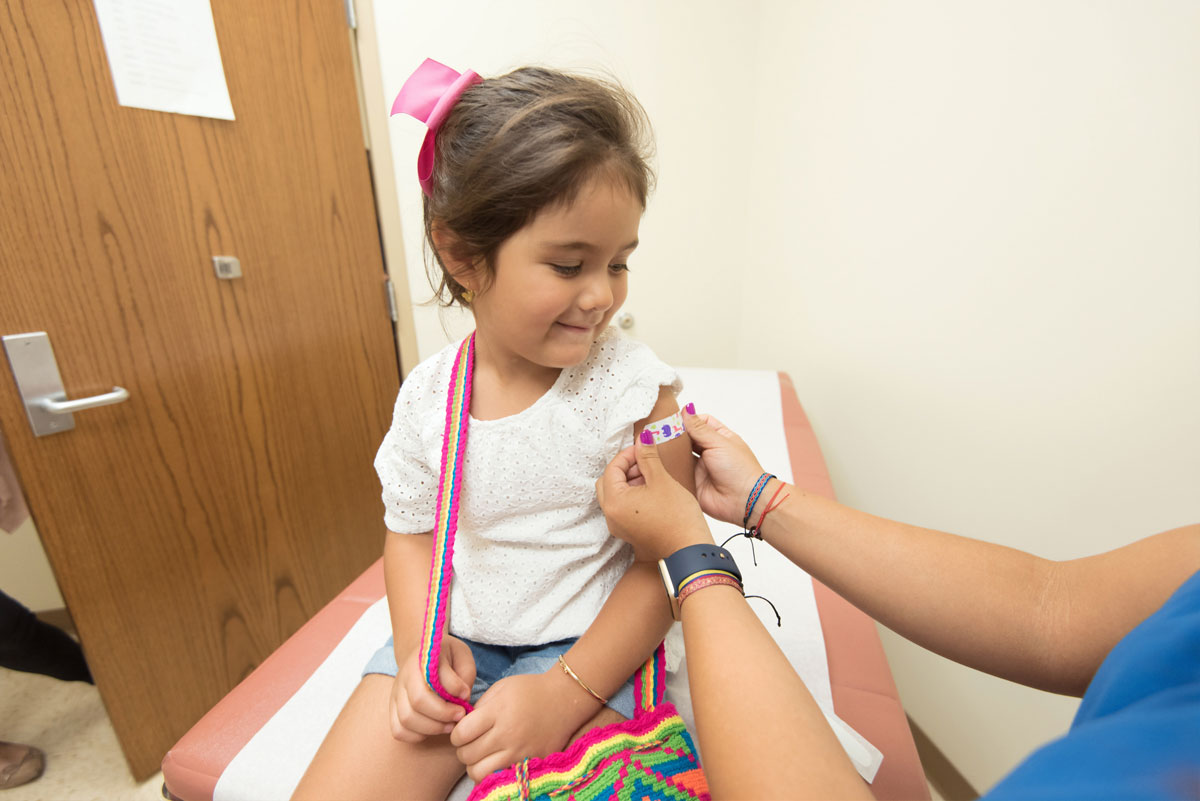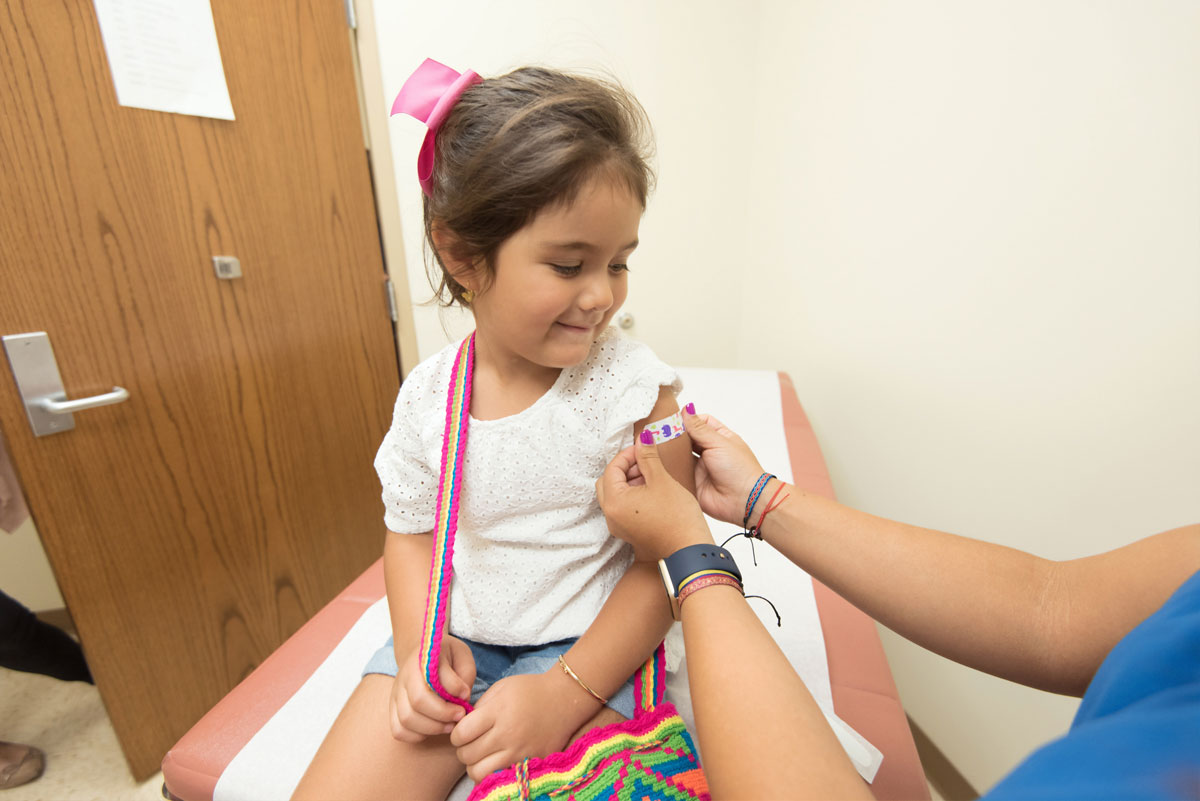
Late on Friday, the FDA approved the Pfizer vaccine for emergency use for children ages 5 to 11. The CDC still needs to sign off on this and provide further guidance, but it’s widely expected that vaccines will be available for children sometime next week. We heard a little over a week ago that the White House had a rollout plan ready to implement as soon as the vaccine received EUA. The amount given to children will be a third of the adult dose and will be packaged differently, in vials with orange tops. A second dose will be given three weeks after the first. Here’s more, from The Guardian.
The US Food and Drug Administration (FDA) on Friday paved the way for children ages five to 11 to get Pfizer’s Covid-19 vaccine.
After the FDA cleared kid-size doses – a third of the amount given to teens and adults – for emergency use, up to 28 million more American children could be eligible for vaccinations as early as next week.
One more regulatory hurdle remains: on Tuesday, advisers to the Centers for Disease Control and Prevention (CDC) will make more detailed recommendations on which children should get vaccinated, with a final decision by the CDC director expected shortly afterwards.
“With this vaccine, kids can go back to something that’s better than being locked at home on remote schooling, not being able to see their friends,” said Dr Kawsar Talaat of Johns Hopkins University. “The vaccine will protect them and also protect our communities.”
A few countries have begun using other Covid-19 vaccines on children under 12, including China, which just began vaccinations for three-year-olds. But many that use the vaccine made by Pfizer and its partner BioNTech are watching the US decision. European regulators just began considering the companies’ kid-size doses.
With FDA’s action, Pfizer plans to begin shipping millions of vials of the pediatric vaccine – in orange caps to avoid mix-ups with the purple-capped doses for everyone else – to doctors’ offices, pharmacies and other vaccination sites. Kids will get two shots, three weeks apart.
The Guardian reported that some members of the FDA advisory panel thought that the vaccine doesn’t need to be given to all children and that it should be targeted at children at higher risk. Very rare side effects, such as heart inflammation, were not found, but it was emphasized that the study might not have been large enough. Hopefully the CDC will recommend the vaccine for all children in this age range. A new study found that only 27% of parents of 5 to 11-year-olds will get their children vaccinated when it’s available, which is disheartening on so many levels. We need school covid vaccination requirements like we have for other vaccines. I drove two and a half hours on a weeknight to get my son vaccinated as soon as he was eligible. My son’s life and health were at stake.
Shame on Newsweek for this trash cover.
Newsweek thought that magazine cover was a good idea? Why didn’t they just add a foot-long needle to the cover for good measure? I mean seriously, give me something to work with here. I feel like I spend half my day lately doing damage control
Many thanks @gorskon for heads up https://t.co/nx36NMm2Ii
— Prof Peter Hotez MD PhD (@PeterHotez) October 29, 2021
Photos Credit: CDC, Miikka Luotio and Kelly Sikkema on Unsplash and Avalon.red